Fatty Acid, Triglyceride, Phospholipid Synthesis and Metabolism. Oxidation of fats involves the reduction of FAD and NAD+. Synthesis of fats involves the oxidation of NADPH.
However, the essential chemistry of the two processes. Both oxidation and synthesis of fats utilize an activated two carbon intermediate, acetyl- Co. A. Like other. enzymes that transfer CO2 to substrates, ACC requires a.
Energy metabolism is the general process by which living cells acquire and use the energy needed to stay alive, to grow, and to reproduce. How is the energy released. It is widely accepted that obesity and associated metabolic diseases, including type 2 diabetes, are intimately linked to diet. However, the gut microbiota has also. Metabolism involves a vast array of chemical reactions, but most fall under a few basic types of reactions that involve the transfer of functional groups of atoms and.
Acetyl- Co. A carboxylase is called an. ABC enzyme due to the requirements for ATP. Biotin, and CO2 for the reaction. This conformational change is enhanced by citrate and inhibited by.
ACC is also controlled through hormone mediated phosphorylation (see below). The acetyl groups that are the products of fatty acid oxidation are linked to Co. ASH. As you should recall. Co. A contains a phosphopantetheine group coupled to AMP. The carrier of acetyl groups (and elongating acyl groups).
The Role of Diet and Exercise for the Maintenance of Fat-Free Mass and Resting Metabolic Rate During Weight Loss. The final destination of a journey is not, after all, the last item on the agenda, but rather some understanding, however simple or provisional, of what one has seen. Metabolism is a term that is used to describe all chemical reactions involved in maintaining the living state of the cells and the organism. Metabolism can be.
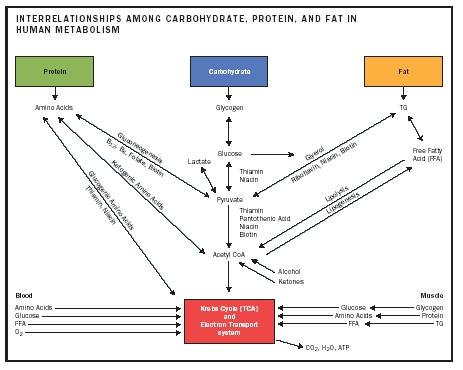
The lipid synthesis and metabolism page provides a detailed description of the structure, synthesis and metabolism of fatty acids, triglycerides, and phospholipids. Three Products That Help Replenish Electrolytes On A Low Carbohydrate Ketogenic Diet Posted on Tuesday, August 7th, 2012 at 3:56 am. By Michael O'Neill. Disorders of carbohydrate metabolism occur in many forms. The most common disorders are acquired. Acquired or secondary derangements in. Read more about a ketogenic diet & the paleo diet. Browse The Paleo Diet
The carrier portion of the synthetic complex is called acyl carrier protein, ACP. The acetyl- Co. A and malonyl- Co.
A are transferred to ACP by. Co. A transacylase and malonyl- Co. A transacylase). The attachment of these carbon. ACP allows them to enter the fatty acid synthesis cycle.
The synthesis of fatty acids from acetyl- Co. A and malonyl- Co.
A is carried out by fatty acid synthase, FAS. The active FAS enzyme exists as a head- to- tail homodimer. All of the reactions of fatty acid synthesis are carried out by the multiple enzymatic activities of FAS. Like fat oxidation, fat synthesis involves four primary enzymatic activities. These are (in order of reaction), . The two reduction reactions require NADPH oxidation to NADP+.
The domain that is required for attachment and transfer of acetyl- Co. A and malonyl- Co. A (acyltransferase domain) is composed of amino acids 4. The phosphopantetheine arm of FAS is attached to a domain composed of amino acids 2.
The primary fatty acid synthesized by FAS is palmitic acid (palmitate). Palmitate is then released from the enzyme via the thioesterase activity of FAS (contained in a domain composed of amino acids 2.
Once released, palmitate can then undergo separate elongation and/or unsaturation to yield other fatty acid molecules. Reactions of fatty acid synthesis catalyzed by fatty acid synthase, FAS. Only half of the normal head- to- tail. FAS is shown. Synthesis of malonyl- Co. A from CO2 and acetyl- Co. A is carried out by ACC as. FAS is initially activated by the incorporation of the acetyl group from acetyl- Co.
A. The acetyl group is initially attached to the. FAS (ACP- SH). This is catalyzed by malonyl/acetyl- Co. A ACP. transacetylase (1 and 2; also called malonyl/acetyltransferase, MAT). This activating acetyl group represents the omega (. Following transfer of the activating acetyl group to a cysteine sulfhydryl in the . The acetyl group attacks the methylene group of the malonyl attached to ACP- SH catalyzed .
The resulting 3- ketoacyl group then undergoes a series of three reactions catalyzed by the . This results in a saturated four carbon (butyryl) group attached to the ACP- SH. This butyryl group is then transferred to the CYS- SH (8) as for the case of the activating acetyl group. At this point another malonyl group is attached to the ACP- SH (3b) and the process begins again. Reactions 4 through 8 are repeated another six times, each beginning with a new malonyl group being added. At the completion of synthesis the saturated 1.
FAS (palmitoyl ACP thioesterase) located in the C- terminal end of the enzyme. Not shown are the released Co. ASH groups. back to the top. Origin of Cytoplasmic Acetyl- Co. AAcetyl- Co. A is generated in the mitochondria primarily from two sources, the. PDH) reaction and fatty acid oxidation.
Under these conditions. Acetyl- Co. A enters the cytoplasm in the form of citrate via the tricarboxylate transport system (see Figure). The resultant oxaloacetate is converted to malate by malate dehydrogenase (MDH). Pathway for the movement of acetyl- Co.
A units from within the mitochondrion to the cytoplasm. Transport of pyruvate across the plasma membrane is catalyzed by the SLC1. A1 protein (also called the. MCT1) and transport across the outer mitochondrial membrane involves a voltage- dependent porin transporter.
Transport across the inner mitochondrial membrane requires a heterotetrameric transport complex (mitochondrial pyruvate carrier) consisting of the MPC1 gene and MPC2 gene encoded proteins. The malate produced by this pathway can undergo oxidative decarboxylation by cytoplasmic malic enzyme. The co- enzyme for this. NADP+ generating NADPH. The advantage of this series of reactions for converting mitochondrial.
Co. A into cytoplasmic acetyl- Co. A is that the NADPH produced by the malic enzyme reaction can be a major source. Humans express three malic enzymes, one cytoplasmic that requires NADP+ and two mitochondrial enzymes, one that requires NADP+ and one that requires NAD+. The cytoplasmic enzyme is called malic enzyme 1 and is encoded by the ME1 gene that is located on chromosome 6q. The NAD+- dependent mitochondrial enzyme is called malic enzyme 2 and is encoded by the ME2 gene located on chromosome 1. RNAs. The NADP+- dependent mitochondrial enzyme is called malic enzyme 3 and is encoded by the ME3 gene located on chromosome 1.
RNAs that all encode the same 6. The role of the mitochondrial malic enzymes is principally to provide the cell with an alternate source of pyruvate under conditions where glycolytic flux in reduced. In these circumstances, the pyruvate generated by the actions of ME2 and/or ME3 come from fumarate precursors such as glutamine. In neurons, as well as in numerous types of tumor cells, mitochondrial malic enzymes allow for the utilization of the amino acid glutamine as a fuel source.
When glutamine is de- aminated by glutaminase the resulting glutamate can also be de- aminated by glutamate dehydrogenase yielding 2- oxoglutarate (. The malate can then be decarboxylated to pyruvate via mitochondrial malic enzyme.
The pyruvate can then be decarboxylated by the PDHc and the resulting acetyl- Co. A can enter the TCA cycle ultimately allowing for glutamine carbons to be oxidized for ATP synthesis. Indeed, this process is energetically equal to glucose- stimulated insulin secretion (GSIS). Regulation of Fatty Acid Metabolism. Regulation by Acetyl- Co. A Carboxylase: ACCOne must consider the global organismal energy requirements in order to effectively understand how the synthesis.
The blood is the carrier of. VLDLs and chylomicrons, fatty acids bound to albumin, amino acids, lactate. The pancreas is the primary organ involved in sensing the organisms dietary and. In response to low blood glucose, glucagon is secreted. The regulation of fat metabolism occurs via. One is short term regulation which is regulation effected by events such as substrate availability, allosteric.
ACC is the rate- limiting (committed) step in fatty. There are two major isoforms of ACC in mammalian tissues. These. are identified as ACC1 (also called ACC. The ACC1. gene (symbol: ACACA) is located on chromosome 1. RNAs that generate proteins from 2. Transcriptional regulation of ACACA is effected.
PI, PII, and PIII), which are located upstream of exons 1, 2, and 5. A, respectively. The presence of the alternatively spliced. ACC1 protein which starts from an AUG present in exon 5.
ACC2 was originally. ACC2 has an N- terminal. I (CPT I) allowing for rapid regulation of. CPT I by the malonyl- Co. A produced by ACC.
Both isoforms of ACC are. Co. A and other. short- and long- chain fatty acyl- Co. As. Citrate triggers the polymerization of. ACC1 which leads to significant increases in its activity. Although ACC2 does. Glutamate and other. ACC isoforms. ACC activity can also be.
Both ACC1 and ACC2 contain at least eight sites. The sites of phosphorylation in ACC2 have not been.
ACC1. Phosphorylation of ACC1 at three serine. S7. 9, S1. 20. 0, and S1. AMPK. leads to inhibition of the enzyme.
AMPK also phosphorylates and inhibits the activity of ACC2. Glucagon- stimulated increases in. AMP and subsequently to increased PKA. ACC where ACC2 is a better substrate for. PKA than is ACC1. Epinephrine activation of .
The activating effects of insulin on ACC are complex and not. It is known that insulin leads to the dephosphorylation of. ACC1 that are AMPK targets in the heart enzyme. This. insulin- mediated effect has not been observed in hepatocytes or adipose tissues. At least a portion of the activating effects of insulin are related to.
AMP levels. Early evidence has shown that phosphorylation and. ACC occurs via the action of an insulin- activated kinase.
However. contradicting evidence indicates that although there is insulin- mediated. ACC this does not result in activation of the enzyme. Indeed, MCD is involved in regulating malonyl- Co. A levels. in multiple tissues. MCD is found in the cytosol, the mitochondria, and in peroxisomes. The MCD enzyme is encoded by the MLYCD gene located on chromosome 1.
Inhibition of MCD results in reduced rates of fatty acid. As well, MCD inhibition. Conversely, inactivation of the MCD gene in mice protects the animals. Additionally, chemical inhibition.
MCD leads to reduced macrophage- associated inflammation in conditions of. The primary mechanism for this effect appears to be via the. PPAR. Transcriptional regulation of the MCD gene is exerted by.
PPAR. Hypothalamic PPAR. The potential therapeutic benefits. MCD activity in the treatment of obesity and diabetes are currently. Additional Regulatory Processes. Control of a given pathways' regulatory enzymes can also occur by alteration of enzyme synthesis and turn- over rates.
Insulin stimulates ACC and FAS synthesis, whereas, starvation. Adipose tissue lipoprotein lipase levels also are increased by.
However, in contrast to the effects of insulin and starvation on adipose tissue. This allows the heart to absorb any available. Starvation also leads to increases. FAS and related enzymes of synthesis. Adipose tissue contains hormone- sensitive lipase (HSL), that is activated by PKA- dependent phosphorylation.
 RSS Feed
RSS Feed
